Essential Safety and Handling Procedures for GLP-1 Receptor Agonist Handling Instructions
Introduction
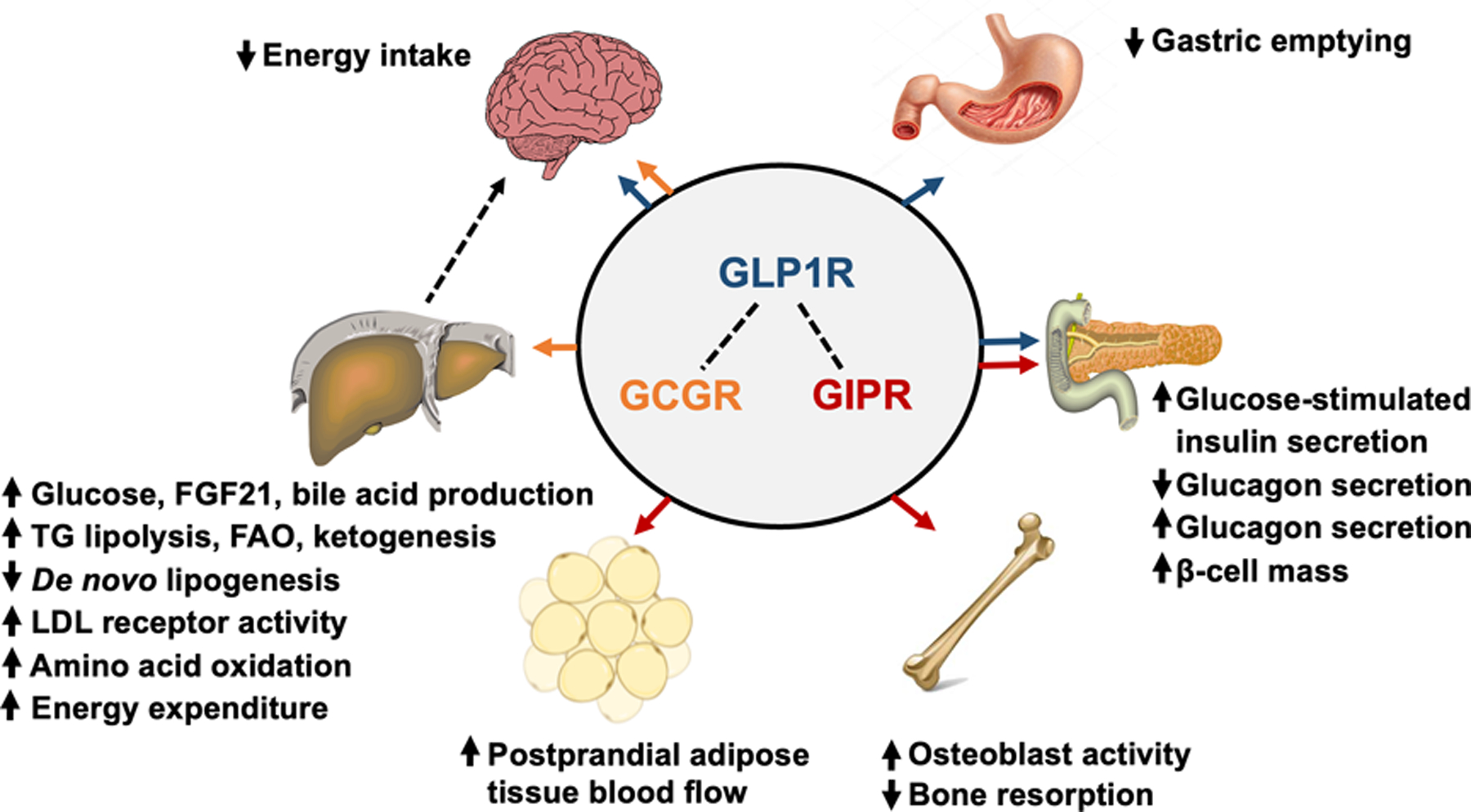
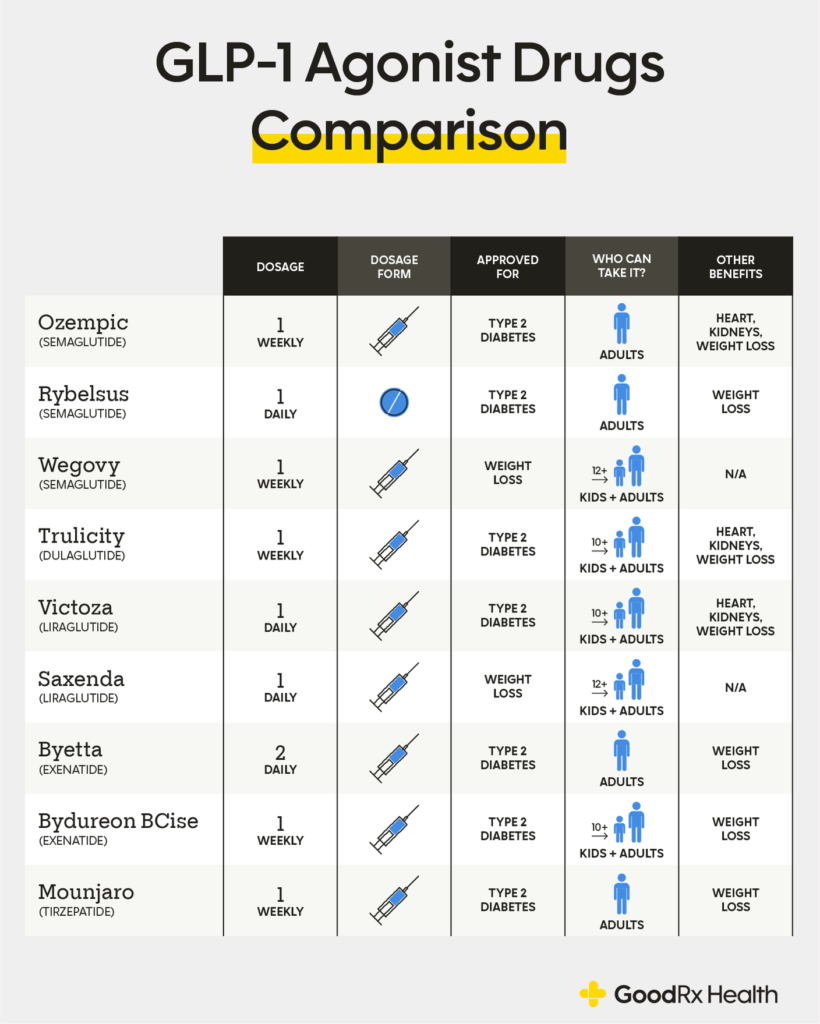
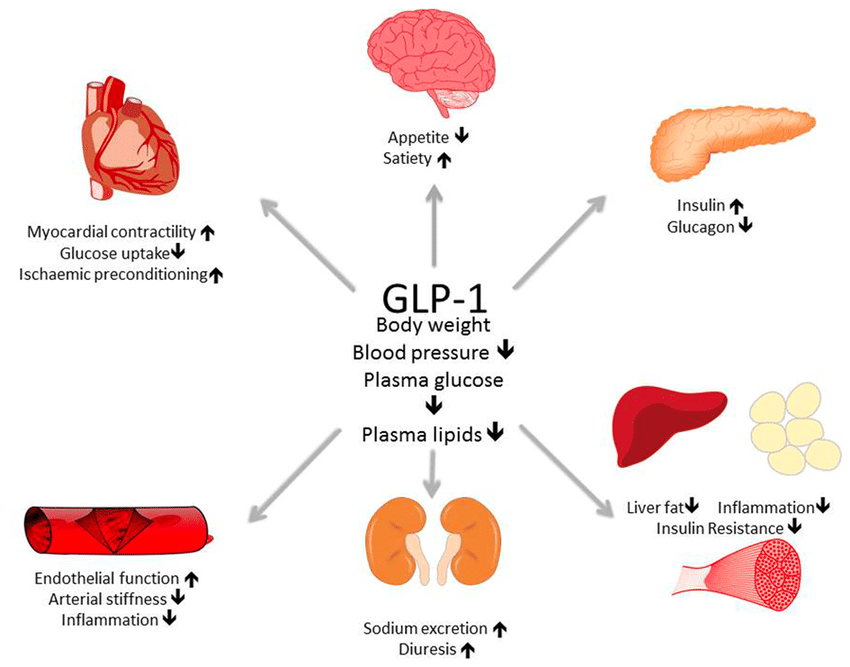
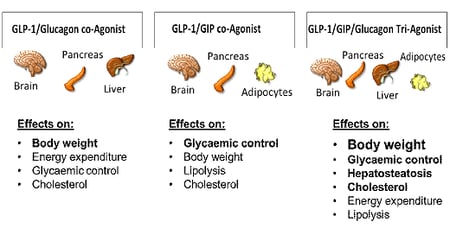
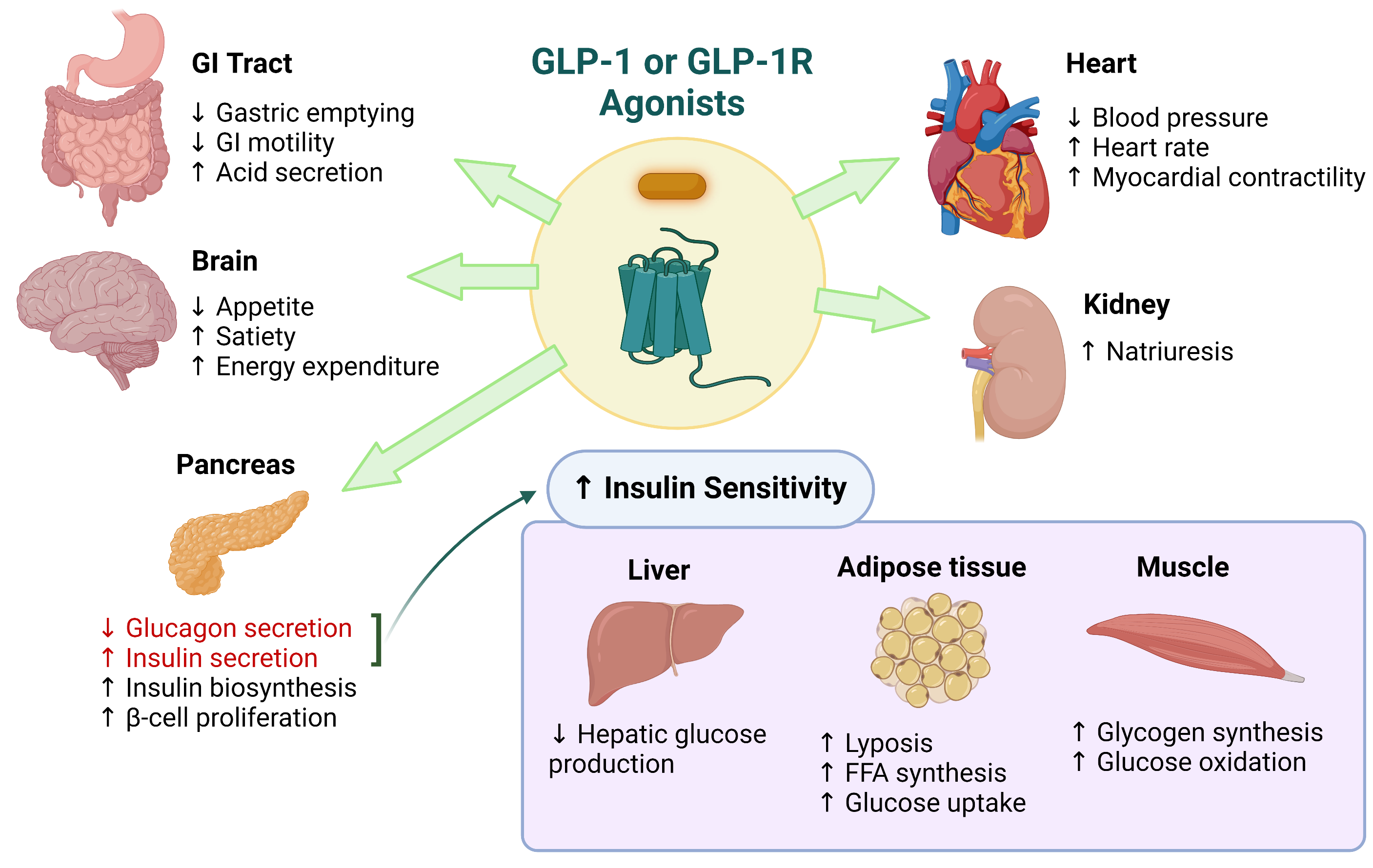
Glucagon-like peptide-1 (GLP-1) receptor agonists are a class of potent medications commonly used to manage type 2 diabetes and support weight loss. These medications are administered via subcutaneous injection, either through a prefilled pen or a syringe and vial. Handling GLP-1 receptor agonists requires proper training, equipment, and adherence to established safety protocols to ensure personal safety and regulatory compliance.
Storing Prefilled Pens
When handling prefilled pens, follow these essential storage procedures:
- Store unused pens in the original box in the medication fridge at 2 to 8°C until the manufacturer's expiry date.
- Label the pens with the expiration date to prevent accidental usage.
- Store pens away from direct sunlight, heat sources, and moisture.
Handling and Administration
For safe administration, follow these guidelines:
- Wash hands thoroughly with soap and water before handling the medication.
- Inspect the pen for any visible damage or tampering.
- Remove the pen from the refrigerator and let it sit at room temperature for a maximum of 40 hours.
- Use a new needle for each injection.
- Administer the medication as prescribed by the healthcare provider.
Disposal Procedures
Proper disposal of GLP-1 receptor agonist pens and needles is crucial to prevent accidental exposure and ensure regulatory compliance:
- Dispose of used pens and needles in puncture-resistant containers.
- Follow local regulations for disposal of biohazardous materials.
- Consider recycling programs for used needles and pens.
Regulatory Compliance and Education
Researchers, scientists, and healthcare professionals handling GLP-1 receptor agonists must adhere to regulatory guidelines and education to ensure safe handling and administration:
- Stay updated on regulatory changes, recommendations, and guidelines.
- Participate in continued education and training programs on GLP-1 receptor agonist handling and administration.
- Participate in medical conferences, workshops, and online courses to stay informed.
Conclusion
GLP-1 receptor agonist handling instructions require attention to safety and regulatory protocols to prevent accidental exposure and ensure personal safety. By following these guidelines and staying informed about regulatory changes, researchers, scientists, and healthcare professionals can provide safe and effective treatment for patients managing type 2 diabetes and obesity.